Not just Aroclors – The Inadvertent PCB Problem
Polychlorinated biphenyls (PCBs) are chlorinated organic compounds widely used in various industrial applications from the 1930s until their ban in the late 1970s. The compounds have excellent heat resistance and electrical insulating properties, so they were commonly used in industrial equipment. PCBs were also prevalent in caulking as a plasticizer in buildings constructed between 1950 and 1979. PCBs do not degrade naturally and can be found in many parts of the environment. They easily bioaccumulate and have been linked to reproductive and developmental problems, immune system suppression, and increased risk of certain cancers.
Biphenyl’s chemical structure is two benzene rings connected by a single covalent bond. In PCBs, one to ten hydrogen atoms in biphenyl are replaced with chlorine atoms. 209 different configurations can occur in this manner, referred to as PCB congeners.
Monsanto Company, a major chemical manufacturer, developed and marketed PCB congener mixtures under the trade name Aroclor starting in the 1930s, each mixture identified by a unique number. The last two digits of these numbers refer to the percentage of chlorine by mass in the mixture. For instance, Aroclor 1254 and Aroclor 1260 were typical in electrical transformers, capacitors, and hydraulic systems. Today’s most common methods for analyzing PCBs in laboratories quantify PCB content based on Aroclor identification using dual-column GC/ECD analysis.
This approach is problematic when non-Aroclor PCBs are most prevalent. Now that the direct manufacture of PCBs is banned, Aroclor PCB mixtures are no longer mass-produced. However, the formulation of pigments and paints has been shown to produce PCB congener byproducts that are not being regularly monitored. These are referred to as inadvertent PCBs (iPCBs). One example of iPCB production is the synthesis of titanium dioxide, which, during a chloride purification process, can produce highly chlorinated PCB congeners as a reaction product [1]. Less chlorinated PCB congeners, such as PCB-11, are formed as impurities in yellow and orange dyes that consumers can be exposed to [2]. Many other routes of iPCB production are being discovered, with a recent study estimating that the amount of iPCBs produced today could very well exceed the amount of Aroclors produced in the 1970s [3].
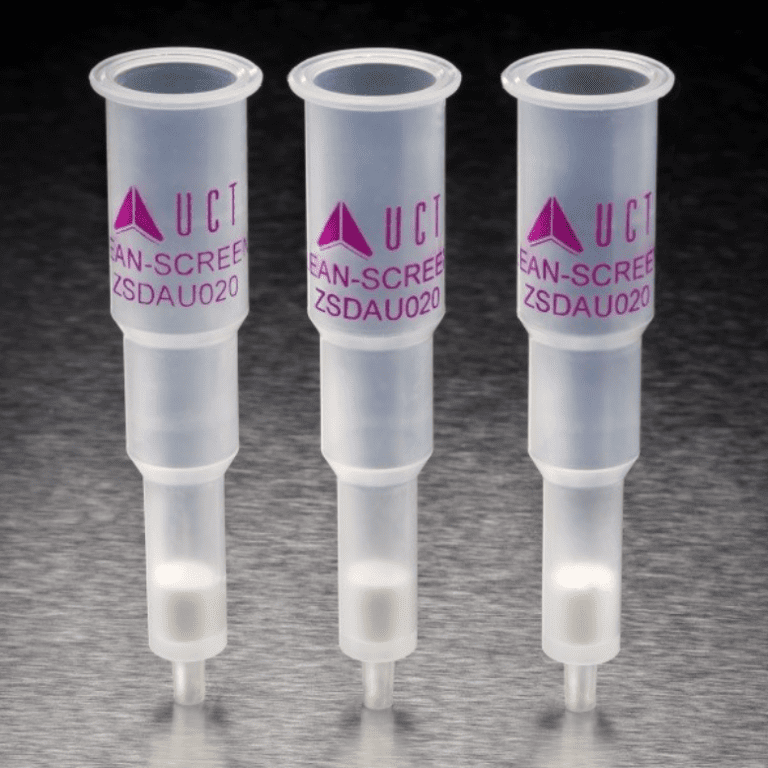
As more studies emerge, iPCBs become a growing concern for environmental safety and human health, requiring regulation’s to be put in place to monitor these contaminants. Testing for iPCBs is possible using gas chromatography techniques such as GC/ECD, GC/MS-SIM, or GC/HRMS. In July 2021, EPA Released Method 1628 for PCB congeners by GC/MS-SIM [4]. This method allows the determination of all 209 PCB congeners. When working with aqueous samples, UCT’s highly crosslinked DVB phase (ECHLD156-P) can be used for solid phase extraction following the below procedure:
A) Acidify the sample to pH 2
B) Attach sample transfer tubes (VMFSTFR12) to the top of the SPE cartridges (ECHLD156-P) and attach the SPE cartridges to an SPE manifold.
C) Wash the SPE cartridges (with transfer tubes connected) using 10 mL methylene chloride, let the solvent soak sorbent for 2 minutes before drawing it to waste. Apply full vacuum pressure for 1 minute.
D) Condition the SPE cartridges with 10 mL methanol, leaving a thin layer above the frit.
E) Equilibrate the SPE cartridges with 15 mL DI water, leaving a thin layer above the frit.
F) Place the stainless-steel end of the transfer tube directly into the sample bottle.
G) Adjust the vacuum pressure so that the flow rate is approximately 5-10 mL/min.
H) When the entire sample has passed through the cartridge, rinse the inside of the sample bottle with 10 mL of reagent water.
I) Dry the SPE cartridges under full vacuum pressure (10-15 in. Hg) for 10 min.
J) Insert glass vials* (40 or 50 mL) into the SPE manifold to collect the eluate.
K) Rinse the sample bottles with 10 mL of 1:1 acetone: n-hexane solution and add the bottle rinse to the SPE cartridges.
L) Let the elution solvent soak the sorbent for 1 minute before drawing it slowly into the collection vial.
M) Twice, repeat the elution step above with 10 mL of 1:9 acetone: n-hexane solution.
N) Concentrate the extracts to 300µL under a gentle stream of nitrogen and reconstitute them to 1mL with hexane.
O) Transfer the extracts to autosampler vials for GC/MS analysis.
[1]Rodenburg, L.A. (2012). Inadvertent PCB production and its impact on water quality. ECOS Annual Meeting, Colorado Springs, CO, 28 August 2012. http://srrttf.org/wpcontent/uploads/2012/08/Lisa-Rodenburg-Slideshow.pdf.
[2]Shang, H., Li, Y., Wang, T., Wang, P., Zhang, H., Zhang, Q., Jiang, G., 2014. The presence of polychlorinated biphenyls in yellow pigment products in China with emphasis on 3,3′-dichlorobiphenyl (PCB 11). Chemosphere 96, 44–50.
[3]David Megson, Ifeoluwa Grace Idowu, Courtney D. Sandau (2024). Is current generation of polychlorinated biphenyls exceeding peak production of the 1970s?, Science of The Total Environment, 2024. https://doi.org/10.1016/j.scitotenv.2024.171436.
[4] Method 1628 Polychlorinated Biphenyl (PCB) Congeners in Water, Soil, Sediment, Biosolids, and Tissue by Low-resolution GC/MS using Selected Ion Monitoring, USEPA, 2021 https://www.epa.gov/system/files/documents/2021-07/method-1628_pcb-congeners-by-low-resolution-gc-ms_jµLy-2021.pdf